China will require export permits for some graphite products, including the spherical graphite used by automakers, beginning 1 December 2023. China “refines more than 90% of the world’s graphite into the material used in almost all EV battery anodes” (the negatively charged portion of a battery), so its decision to reduce exports presents a major risk for the car industry.(1)
Companies outside of China have long been concerned about the security of supply of industrial-grade carbon products. That concern has led to universities and companies expediting their research into alternatives to traditional products.
One of the most exciting areas of active development is using novel catalysts to produce high-quality carbon products from methane gas without emitting greenhouse gases.
SEVERAL COMPANIES WORK WITH METHANE SPLITTING
Forbes recently published an interesting article about a dozen energy and technology companies working quietly to produce high-value carbon products for the electric vehicle market using natural gas and a methane pyrolysis production pathway.(2) We are one of those companies, and now is the time to stop being quiet.
Speaking strictly scientifically, methane pyrolysis is a process that uses heat to split methane (CH₄) into hydrogen (H₂) and solid carbon. However, without catalysts, methane pyrolysis only occurs at temperatures above 1100–1200°C.(3) Due to the high energy demand, the non-catalytic process is not economically viable for large-scale industrial needs.
Catalysts are the magic element all the companies in this space are researching. Incorporating a catalyst for methane splitting can significantly reduce the reaction temperature. Several metals and non-metal catalysts have been used to meet companies’ goals for methane-splitting technologies, our preferred term to avoid confusion with the non-catalyst-enhanced methane pyrolysis process.
As we develop this technology, we turn side streams into revenue streams. Many companies have developed methane splitting as a means to produce emission-free hydrogen, so-called turquoise hydrogen, and carbon has been a side product. Methane splitting still yields low-carbon hydrogen, and the carbon it creates has proven to be much more than just insignificant black powder that must be stored somewhere away from the atmosphere.
CARBON PRODUCTS CAN BE TAILORED
Lower reaction temperatures and thus lower energy requirements is only one of the benefits of using suitable catalysts. Catalysts can also influence the quality of the resulting solid carbon. For instance, by using a suitable catalyst family in carbon production, companies can produce a mixture of nanocarbons whose properties are suitable for vehicle batteries, electrodes and supercapacitors. This can be seen in Figure 1 presenting the electrical conductivity of a carbon product made by splitting methane with a special catalyst . Other catalysts produce simpler forms of carbon that customers can use, for instance, in the steel industry.
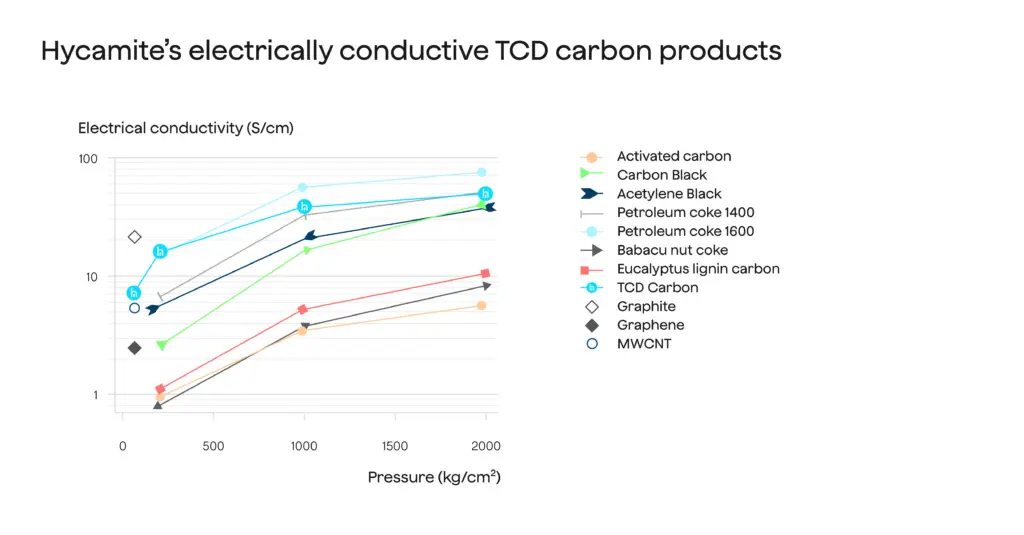
igure 1: Carbon product from methane splitting can show electric conductivity.
After following how our research team can tailor new catalysts to customers’ needs, even managing to create value from industrial side streams that had previously been considered problematic waste, I am excited to see what other research teams around the globe will be able to do.
There are still other reasons why several companies are now eyeing methane as a source of urgently needed carbon products.
PRODUCTION CAN BE SCALED QUICKLY AND CLOSE TO USE SITES
Methane splitting is a solution that can be scaled quickly. Speed is crucial now that Chinese restrictions are entering into force this year – not in the distant future.
Methane is available all around the world. The methane trade has a well-established market system with infrastructure and regulations.
If natural gas is unavailable as a methane source, biogas can be used instead. Using biomethane creates carbon sinks when the captured carbon is permanently fixed in batteries and other products rather than released into the atmosphere. As shown in Figure 2, the carbon sink size can be significant.
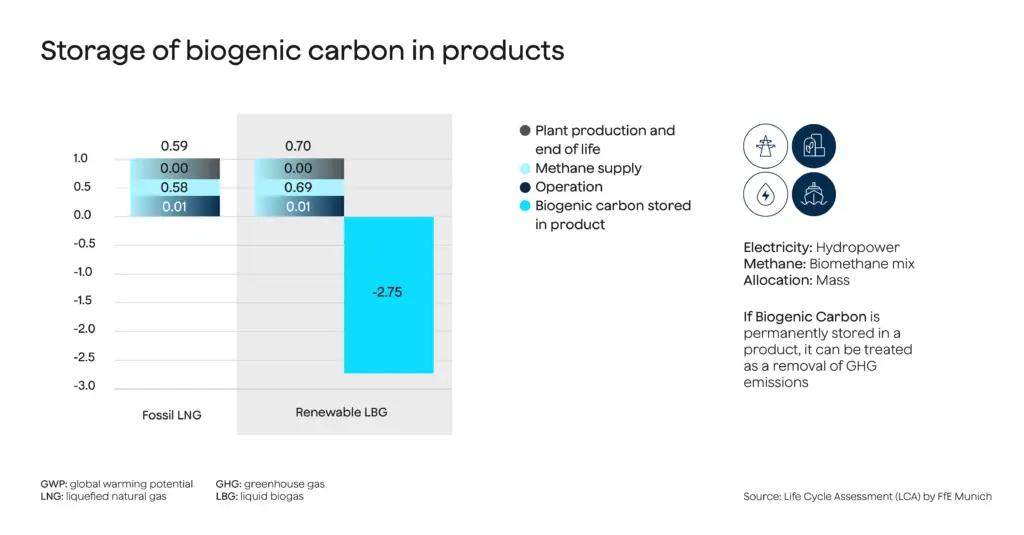
Figure 2: When using biomethane, methane-splitting technologies can create carbon sinks.
Methane can also be split in relatively small production units next to the industrial plants where the carbon products are used. Doing so not only increases the security of supply but also makes it easier to invest in production of precisely the right scale and tailor the production to each customer’s needs.
PROCESS WITHOUT GREENHOUSE GAS EMISSIONS
Methane splitting is far more environmentally sound than synthetic carbon production could ever be. To break a methane molecule apart, methane gas is constantly heated without oxygen. After the methane molecule has been split, the carbon is immediately collected in a solid form. There is no room in the process for carbon dioxide formation, and thus carbon dioxide cannot leak into the atmosphere even by accident.
We are sometimes criticised for the methane emissions that occur upstream of our process. I absolutely agree that when using methane for any reason, upstream emissions should be not only minimised but also wholly prevented.
Sultan bin Ahmed Al Jaber, UAE Minister of Industry and High Technology and President of Cop28, recently made developing plans to eliminate the oil and gas industry’s methane emissions by 2030 a top priority for Cop28. Al Jaber said that more than 20 oil and gas companies have committed to end methane emissions by 2030, and more have shown interest in joining this initiative. I support this initiative. We need methane for several purposes, but the production and use of methane should not harm the planet.
Niina Grönqvist, VP, Carbon Products of Hycamite
This article was originally published on LinkedIn, read the original article here.
(1)https://www.reuters.com/business/autos-transportation/chinas-graphite-curbs-will-accelerate-plans-around-alternatives-2023-10-20/
(2)https://www.forbes.com/sites/dipkabhambhani/2023/11/01/turquoise-hydrogen-producers-could-capture-flourishing-graphite-market/
(3)https://onlinelibrary.wiley.com/doi/10.1002/cite.202000029





